TATA结合蛋白
位於6號人類染色體的基因
TATA结合蛋白质(英語:TATA-binding protein,TBP)是一种通用转录因子,具体结合于DNA序列上的TATA盒。TATA盒位于真核基因的转录起始位点上游约25个bp,属于啟動子区[6]。TBP可以和各个TBP相关因子共同组成通用转录因子TFIID,再进一步组成RNA聚合酶Ⅱ的转录起始复合物[7]。
| TBP | |||||||||
|---|---|---|---|---|---|---|---|---|---|
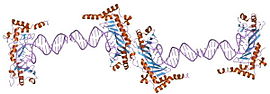 crystal structure of a yeast brf1-tbp-dna ternary complex | |||||||||
| 鑑定 | |||||||||
| 標誌 | TBP | ||||||||
| Pfam | PF00352(旧版) | ||||||||
| Pfam宗系 | CL0407(旧版) | ||||||||
| InterPro | IPR000814 | ||||||||
| PROSITE | PDOC00303 | ||||||||
| SCOP | 1tbp / SUPFAM | ||||||||
| |||||||||
蛋白质的相互作用
编辑TATA结合蛋白与下列蛋白質进行互作:
- BRF1,[8][9]
- BTAF1,[10][11]
- C-Fos,[12]
- C-jun,[13]
- EDF1,[14][15][16]
- GTF2B (TFIIB),[17][18]
- GTF2A1 (TFIIA subunit 1),[17][19][20][21]
- GTF2F1 (TFIIF subunit 1)[10][22][23]
- GTF2H4 (TFIIH subunit 4),[10]
- Mdm2,[24][24][25]
- MSX1,[26][27][28]
- NFYB,[29]
- P53,[30][31]
- PAX6,[31]
- POLR2A,[10]
- POU2F1,[32]
- RELA,[33][34]
- NR2B1,[35]
- TAF1,[29][36][37][38]
- TAF4,[39]
- TAF5,[29][37][40]
- TAF6,[29][37][39]
- TAF7,[29][39]
- TAF9.[29][41]
- TAF10,[29][37]
- TAF11,[29][42][43]
- TAF13,[42]
- TAF15.[44]
参考文献
编辑- ^ 與TATA结合蛋白相關的疾病;在維基數據上查看/編輯參考.
- ^ 2.0 2.1 2.2 GRCh38: Ensembl release 89: ENSG00000112592 - Ensembl, May 2017
- ^ 3.0 3.1 3.2 GRCm38: Ensembl release 89: ENSMUSG00000014767 - Ensembl, May 2017
- ^ Human PubMed Reference:. National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Mouse PubMed Reference:. National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ The molecular basis of eukaryotic transcription. Proc. Natl. Acad. Sci. U.S.A. 2007, 104 (32): 12955–61. PMC 1941834 . PMID 17670940. doi:10.1073/pnas.0704138104.
- ^ Transcription of eukaryotic protein–coding genes. Annu. Rev. Genet. 2000, 34: 77–137. PMID 11092823. doi:10.1146/annurev.genet.34.1.77.
- ^ McCulloch V, Hardin P, Peng W, Ruppert JM, Lobo-Ruppert SM. Alternatively spliced hBRF variants function at different RNA polymerase III promoters. EMBO J. August 2000, 19 (15): 4134–43. PMC 306597 . PMID 10921893. doi:10.1093/emboj/19.15.4134.
- ^ Wang Z, Roeder RG. Structure and function of a human transcription factor TFIIIB subunit that is evolutionarily conserved and contains both TFIIB- and high-mobility-group protein 2-related domains. Proc. Natl. Acad. Sci. U.S.A. July 1995, 92 (15): 7026–30. PMC 41464 . PMID 7624363. doi:10.1073/pnas.92.15.7026.
- ^ 10.0 10.1 10.2 10.3 Scully R, Anderson SF, Chao DM, Wei W, Ye L, Young RA, Livingston DM, Parvin JD. BRCA1 is a component of the RNA polymerase II holoenzyme. Proc. Natl. Acad. Sci. U.S.A. May 1997, 94 (11): 5605–10. PMC 20825 . PMID 9159119. doi:10.1073/pnas.94.11.5605.
- ^ Chicca JJ, Auble DT, Pugh BF. Cloning and biochemical characterization of TAF-172, a human homolog of yeast Mot1. Mol. Cell. Biol. March 1998, 18 (3): 1701–10. PMC 108885 . PMID 9488487.
- ^ Metz R, Bannister AJ, Sutherland JA, Hagemeier C, O'Rourke EC, Cook A, Bravo R, Kouzarides T. c-Fos-induced activation of a TATA-box-containing promoter involves direct contact with TATA-box-binding protein. Mol. Cell. Biol. September 1994, 14 (9): 6021–9. PMC 359128 . PMID 8065335. doi:10.1128/MCB.14.9.6021.
- ^ Franklin CC, McCulloch AV, Kraft AS. In vitro association between the Jun protein family and the general transcription factors, TBP and TFIIB. Biochem. J. February 1995, 305 (3): 967–74. PMC 1136352 . PMID 7848298. doi:10.1042/bj3050967.
- ^ Brendel C, Gelman L, Auwerx J. Multiprotein bridging factor-1 (MBF-1) is a cofactor for nuclear receptors that regulate lipid metabolism. Mol. Endocrinol. June 2002, 16 (6): 1367–77. PMID 12040021. doi:10.1210/mend.16.6.0843.
- ^ Mariotti M, De Benedictis L, Avon E, Maier JA. Interaction between endothelial differentiation-related factor-1 and calmodulin in vitro and in vivo. J. Biol. Chem. August 2000, 275 (31): 24047–51. PMID 10816571. doi:10.1074/jbc.M001928200.
- ^ Kabe Y, Goto M, Shima D, Imai T, Wada T, Morohashi Ki, Shirakawa M, Hirose S, Handa H. The role of human MBF1 as a transcriptional coactivator. J. Biol. Chem. November 1999, 274 (48): 34196–202. PMID 10567391. doi:10.1074/jbc.274.48.34196.
- ^ 17.0 17.1 Tang H, Sun X, Reinberg D, Ebright RH. Protein–protein interactions in eukaryotic transcription initiation: structure of the preinitiation complex. Proc. Natl. Acad. Sci. U.S.A. February 1996, 93 (3): 1119–24. PMC 40041 . PMID 8577725. doi:10.1073/pnas.93.3.1119.
- ^ Bushnell DA, Westover KD, Davis RE, Kornberg RD. Structural basis of transcription: an RNA polymerase II-TFIIB cocrystal at 4.5 Angstroms. Science. February 2004, 303 (5660): 983–8. PMID 14963322. doi:10.1126/science.1090838.
- ^ DeJong J, Bernstein R, Roeder RG. Human general transcription factor TFIIA: characterization of a cDNA encoding the small subunit and requirement for basal and activated transcription. Proc. Natl. Acad. Sci. U.S.A. April 1995, 92 (8): 3313–7. PMC 42156 . PMID 7724559. doi:10.1073/pnas.92.8.3313.
- ^ Ozer J, Mitsouras K, Zerby D, Carey M, Lieberman PM. Transcription factor IIA derepresses TATA-binding protein (TBP)-associated factor inhibition of TBP-DNA binding. J. Biol. Chem. June 1998, 273 (23): 14293–300. PMID 9603936. doi:10.1074/jbc.273.23.14293.
- ^ Sun X, Ma D, Sheldon M, Yeung K, Reinberg D. Reconstitution of human TFIIA activity from recombinant polypeptides: a role in TFIID-mediated transcription. Genes Dev. October 1994, 8 (19): 2336–48. PMID 7958900. doi:10.1101/gad.8.19.2336.
- ^ Ruppert S, Tjian R. Human TAFII250 interacts with RAP74: implications for RNA polymerase II initiation. Genes Dev. November 1995, 9 (22): 2747–55. PMID 7590250. doi:10.1101/gad.9.22.2747.
- ^ Malik S, Guermah M, Roeder RG. A dynamic model for PC4 coactivator function in RNA polymerase II transcription. Proc. Natl. Acad. Sci. U.S.A. March 1998, 95 (5): 2192–7. PMC 19292 . PMID 9482861. doi:10.1073/pnas.95.5.2192.
- ^ 24.0 24.1 Thut CJ, Goodrich JA, Tjian R. Repression of p53-mediated transcription by MDM2: a dual mechanism. Genes Dev. August 1997, 11 (15): 1974–86. PMC 316412 . PMID 9271120. doi:10.1101/gad.11.15.1974.
- ^ Léveillard T, Wasylyk B. The MDM2 C-terminal region binds to TAFII250 and is required for MDM2 regulation of the cyclin A promoter. J. Biol. Chem. December 1997, 272 (49): 30651–61. PMID 9388200. doi:10.1074/jbc.272.49.30651.
- ^ Shetty S, Takahashi T, Matsui H, Ayengar R, Raghow R. Transcriptional autorepression of Msx1 gene is mediated by interactions of Msx1 protein with a multi-protein transcriptional complex containing TATA-binding protein, Sp1 and cAMP-response-element-binding protein-binding protein (CBP/p300). Biochem. J. May 1999, 339 (3): 751–8. PMC 1220213 . PMID 10215616. doi:10.1042/0264-6021:3390751.
- ^ Zhang H, Hu G, Wang H, Sciavolino P, Iler N, Shen MM, Abate-Shen C. Heterodimerization of Msx and Dlx homeoproteins results in functional antagonism. Mol. Cell. Biol. May 1997, 17 (5): 2920–32. PMC 232144 . PMID 9111364. doi:10.1128/mcb.17.5.2920.
- ^ Zhang H, Catron KM, Abate-Shen C. A role for the Msx-1 homeodomain in transcriptional regulation: residues in the N-terminal arm mediate TATA binding protein interaction and transcriptional repression. Proc. Natl. Acad. Sci. U.S.A. March 1996, 93 (5): 1764–9. PMC 39855 . PMID 8700832. doi:10.1073/pnas.93.5.1764.
- ^ 29.0 29.1 29.2 29.3 29.4 29.5 29.6 29.7 Bellorini M, Lee DK, Dantonel JC, Zemzoumi K, Roeder RG, Tora L, Mantovani R. CCAAT binding NF-Y-TBP interactions: NF-YB and NF-YC require short domains adjacent to their histone fold motifs for association with TBP basic residues. Nucleic Acids Res. June 1997, 25 (11): 2174–81. PMC 146709 . PMID 9153318. doi:10.1093/nar/25.11.2174.
- ^ Seto E, Usheva A, Zambetti GP, Momand J, Horikoshi N, Weinmann R, Levine AJ, Shenk T. Wild-type p53 binds to the TATA-binding protein and represses transcription. Proc. Natl. Acad. Sci. U.S.A. December 1992, 89 (24): 12028–32. PMC 50691 . PMID 1465435. doi:10.1073/pnas.89.24.12028.
- ^ 31.0 31.1 Cvekl A, Kashanchi F, Brady JN, Piatigorsky J. Pax-6 interactions with TATA-box-binding protein and retinoblastoma protein. Invest. Ophthalmol. Vis. Sci. June 1999, 40 (7): 1343–50. PMID 10359315.
- ^ Zwilling S, Annweiler A, Wirth T. The POU domains of the Oct1 and Oct2 transcription factors mediate specific interaction with TBP. Nucleic Acids Res. May 1994, 22 (9): 1655–62. PMC 308045 . PMID 8202368. doi:10.1093/nar/22.9.1655.
- ^ Guermah M, Malik S, Roeder RG. Involvement of TFIID and USA components in transcriptional activation of the human immunodeficiency virus promoter by NF-kappaB and Sp1. Mol. Cell. Biol. June 1998, 18 (6): 3234–44. PMC 108905 . PMID 9584164. doi:10.1128/mcb.18.6.3234.
- ^ Schmitz ML, Stelzer G, Altmann H, Meisterernst M, Baeuerle PA. Interaction of the COOH-terminal transactivation domain of p65 NF-kappa B with TATA-binding protein, transcription factor IIB, and coactivators. J. Biol. Chem. March 1995, 270 (13): 7219–26. PMID 7706261. doi:10.1074/jbc.270.13.7219.
- ^ Schulman IG, Chakravarti D, Juguilon H, Romo A, Evans RM. Interactions between the retinoid X receptor and a conserved region of the TATA-binding protein mediate hormone-dependent transactivation. Proc. Natl. Acad. Sci. U.S.A. August 1995, 92 (18): 8288–92. PMC 41142 . PMID 7667283. doi:10.1073/pnas.92.18.8288.
- ^ Siegert JL, Robbins PD. Rb inhibits the intrinsic kinase activity of TATA-binding protein-associated factor TAFII250. Mol. Cell. Biol. January 1999, 19 (1): 846–54. PMC 83941 . PMID 9858607.
- ^ 37.0 37.1 37.2 37.3 Ruppert S, Wang EH, Tjian R. Cloning and expression of human TAFII250: a TBP-associated factor implicated in cell-cycle regulation. Nature. March 1993, 362 (6416): 175–9. PMID 7680771. doi:10.1038/362175a0.
- ^ O'Brien T, Tjian R. Functional analysis of the human TAFII250 N-terminal kinase domain. Mol. Cell. May 1998, 1 (6): 905–11. PMID 9660973. doi:10.1016/S1097-2765(00)80089-1.
- ^ 39.0 39.1 39.2 Pointud JC, Mengus G, Brancorsini S, Monaco L, Parvinen M, Sassone-Corsi P, Davidson I. The intracellular localisation of TAF7L, a paralogue of transcription factor TFIID subunit TAF7, is developmentally regulated during male germ-cell differentiation. J. Cell. Sci. May 2003, 116 (Pt 9): 1847–58. PMID 12665565. doi:10.1242/jcs.00391.
- ^ Tao Y, Guermah M, Martinez E, Oelgeschläger T, Hasegawa S, Takada R, Yamamoto T, Horikoshi M, Roeder RG. Specific interactions and potential functions of human TAFII100. J. Biol. Chem. March 1997, 272 (10): 6714–21. PMID 9045704. doi:10.1074/jbc.272.10.6714.
- ^ Martinez E, Palhan VB, Tjernberg A, Lymar ES, Gamper AM, Kundu TK, Chait BT, Roeder RG. Human STAGA complex is a chromatin-acetylating transcription coactivator that interacts with pre-mRNA splicing and DNA damage-binding factors in vivo. Mol. Cell. Biol. October 2001, 21 (20): 6782–95. PMC 99856 . PMID 11564863. doi:10.1128/MCB.21.20.6782-6795.2001.
- ^ 42.0 42.1 Mengus G, May M, Jacq X, Staub A, Tora L, Chambon P, Davidson I. Cloning and characterization of hTAFII18, hTAFII20 and hTAFII28: three subunits of the human transcription factor TFIID. EMBO J. April 1995, 14 (7): 1520–31. PMC 398239 . PMID 7729427.
- ^ May M, Mengus G, Lavigne AC, Chambon P, Davidson I. Human TAF(II28) promotes transcriptional stimulation by activation function 2 of the retinoid X receptors. EMBO J. June 1996, 15 (12): 3093–104. PMC 450252 . PMID 8670810.
- ^ Hoffmann A, Roeder RG. Cloning and characterization of human TAF20/15. Multiple interactions suggest a central role in TFIID complex formation. J. Biol. Chem. July 1996, 271 (30): 18194–202. PMID 8663456. doi:10.1074/jbc.271.30.18194.





